INTRODUCTION
Common Name: Paclobutrazol 25% SC
Use: Used on fruit trees to inhibit vegetative growth and to improve fruit set; on pot-grown ornamentals and flower crops (e.g. chrysanthemums, begonias, freesias, poinsettias and bulbs) to inhibit growth; on rice to increase tillering, reduce lodging, and increase yield; on turf to retard growth; and on grass seed crops to reduce height and prevent lodging. To be applied as a foliar spray, as a soil drench, or by trunk injection. Has some fungicidal activity against mildew and rusts. Phytotoxicity Non-phytotoxic, though it intensifies greening. Some spotting has been noted on periwinkle foliage at higher temperatures.
Company Name: Penzeal Industry Co., Ltd.
Situation : Asia
Nationality : China
Address : Rm.709 East Block Huatai Zonghe Building, No.76 Xinwen Road, Futian District, Shenzhen, P. R. China
Remarks : Penzeal Industry Co., Ltd. is a specialized formulation factory in the field of agrochemical products. We possess very strong networks of relationships, complete techniques and staffing. We own a very professional team, whose members have been engaged in agrochemical products for more than 10 years. We have established long-term and good co-operation with most outstanding technical factories in China, so we are fully confident to offer products with good quality but low price to our esteemed customers. In terms of shipment, we have very perfect Export service system, and have established good relationship with many shipping and air companies, so that we can choose the best shipping or air lines for your goods to different countries and areas, of course accordingly we offer a series of service including fast custom declaration and insurance.
APPLICANT
Name : Penzeal Industry Co., Ltd.
Address : Rm.709 East Block Huatai Zonghe Building, No.76 Xinwen Road, Futian District, Shenzhen, P. R. China
Nationality : China
Tel. : 86-755-83864316
Authority cert. : We are registered in the Ministry of Agriculture as an Established Company.
Indentity cert. : We are registered in the Ministry of Agriculture as an Established Company.
Remarks : Penzeal Industry Co., Ltd. is a specialized trade company in the field of import & export of agrochemical products. The company possesses very strong networks of relationships, complete techniques and staffing. In respect of products sources, we own a very professional team, whose members have been engaged in purchasing and operation of agrochemical products for more than 10 years. For more, we have established long-term and good co-operation relationship with most outstanding suppliers in China, so we are fully confident to offer products with good quality but low price to our esteemed customers. In terms of shipment, we have very perfect Import & Export service system, and have established good co-operation relationship with many shipping and air companies, so that we can chose the best shipping or air lines for your goods to different countries and areas, of course accordingly we offer a series of service including fast custom declaration and insurance.
Chemistry of The Product
SYNONYMS
Common Name: Paclobutrazol
Code Number: N/A
Trade Name: Alpha Grow 25% SC
Initial Regist.: N/A
Chemical Class: Triazole
Local Synonyms: N/A
Chemical Name:
Source: Paclobutrazol
Structural Formula:
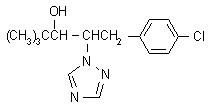
Physical and Chemical Properties:
Molecular Weight: 293.8
Physical Form: Crystalline solid
Technical Purity: 95%
Vapour Pressure: 0.001 mPa (20 oC)
Colour & Odour: White, odourless
Density: 1.22
Solubility: In water 26 mg/l (20 oC). In acetone 110, cyclohexanone 180, dichloromethane 100, hexane 10, xylene 60, methanol 150, propylene glycol 50 (all in g/l, 20 oC).
Octanol Number : logP = 3.2
Type of Formulations And Specifications
CONTENTS |
TYPE OF FORMULATIONS |
Soluble concentrate |
|
|
ACTIVE INGREDIENT
INERT INGREDIENT |
Paclobutrazol 25%
Dispersant 2%, wetting agent, 1%, emulsifier 1%, thickener 0.5%, filler 70.5% |
|
|
SPECIFICATIONS OF FORMULATIONS
Appearance : Off-white suspension
Alkalinity or Acidity : N/A
Suspensibility : 80% min
Emulsification Properties : N/A
Density : 1.06 ± 0.05
Viscosity : N/A
Persistent foam : 25 ml max (after 1 min)
Flash Point : N/A
Freeze Point : N/A
Wettability : N/A.
Particle Size : 98% min pass through 75μm wet sieve .
Compatability : Incompatible with strong acid or alkaline agrochemicals
Heat Stability : After storage at 54 ± 2oC for 14 days, the determined average active ingredient content must not be lower than 90% relative to the determined mean content found before storage and the formulation shall continue to comply with the clauses for suspensibility and wet sieve test.
Storage Stability : Qualified in storage stability test
Mixing Properties : N/A
Methods of Analysis : GC or HPLC
Levels of Impurities : N/A.
Biological Spectrum
Pesticidal efficacy (list of pests):
Inhibit growth, increase tillering, reduce lodging and increasing yield. Also has some fungicidal activity against mildew and rusts.
Mode of action:
Plant growth regulator taken up into the xylem through the leaves, stems, or roots, and translocated to growing sub-apical meristems. Produces more compact plants and enhances flowering and fruiting.
Use and Recommendations:
Target Pest |
CROP |
APPLICTION RATE / Faddan |
APPLICATION METHOD |
Inhibit grow, increase tillering, precent lodging |
Rice |
1.0 - 1.2 g/L water |
Spray |
Inhibit grow, increase tillering, precent lodging |
Wheat |
0.5 - 0.8 g/L water
|
Spray
|
Inhibit grow, precent lodging |
Peanut |
1.5 - 2.0 g/L water |
Spray after flowering stage |
Inhibit branches, increase fruit twig |
Grape |
2 g/L water |
Foliar spray after flowering stage |
Inhibit growth |
Tomato |
0.4 - 0.5 g/L water |
Foliar spray |
Inhibit brancees |
Rape |
0.3 - 0.4 g/L water |
Foliar spray at 3 - 4 leaves stage |
Phytotoxicity:
No phytotoxicity.
PHI:
Rice 30 d, wheat 30 d, peanut 14 d, grape 14 d, tomato 7 d, rape 7 d.
Toxicological Studies
Acute Mammalian Toxicity:
Acute toxicity |
ANIMAL |
LD50 mg/Kg body weight |
Technical |
Form |
Oral
Dermal
Inhaltion (mg/m3/2H) |
Rat
Rabbit
Rat |
1300-2000
>1000
3130-4790 |
>2000
>2000
>4000 |
Symptoms |
EYE: No irritation |
SKIN: No irritation |
WHO
Classification |
Toxicity Category |
Label Signal Word |
High I |
Mod II |
Low III |
Danger |
Warning |
Caution |
Techn.
Form
Impurities |
|
|
III
III
III |
|
|
CAUTION
CAUTION
CAUTION |
Chronic Toxicity
Sprague-Dawley derived Crt:CD (SD) BR rats (60/sex/dose with 10/sex/dose scheduled for sacrifice at 52 weeks) were ded on a diet containing paclobutrazol (92.4% puritty) at 0, 50, 250 or 1250 ppm (approximately 0, 1.4, 6.8 or 33.8 mg/kg-day for males; 0, 1.6, 8.8 or 48.8 mg/kg-day for females; based on reported food consumption) for 104 weeks (Shaw, 1986b). Females exhibited a significant (P<0.05) decrement in body weight gain at doses of 250 and 1250 ppm. At the highest dose (1250 ppm), live weight, adjusted for body weight, were significantly increased (P<0.01) for both males and females. Certrilobular hypertrophy accompanied by steatosis were seen in the livers at the highest dose at both 52 and 104 weeks. At 104 weeks, steatosis was also noted in some males dosed at 250. No major histopathological changes in the liver, such as chlolestasis or necrosis, were reported. No overall treatment effects were observed in glutamate oxaloacetate transaminase (SGOT), glytamate pyruvate transaminase (SGPT), alkaline phosphatase, glucose, cholesterol, total protein, albumin, or albumin/globulin ratio. Serum striglyceride values were significantly reduced compared to controls in females at 250 and 1250 ppm at 39 weeks, and at 1250 ppm at 52 weeks. [The change in the serum triglycerides may indicate an alteration in lipid metabolism which leads to the observed steatosis.] Also, blood urea nitrogen (BUN) was decrement in body weight gain, and decreased serum triglyceride values in femals was 1.6 mg/kg-day at 104 weeks. This study was acceptable according to the Federal Fungicide, Insecticide, and Rodenticide Act(FIFRA) guidelines.
Carcinogenicity (WHO-IARC)
This is a combined chronic feeding oncogenicity study. The study design is acceptable according to the EPA Testing Guidelines. The measurement of serum concentration of electrolytes was not part of the protocol. Since the chronic toxicity of this chemical in rats appears to be minimal, it is not likely that any effects would have been ovserved in these tests. Therefore, in this praticular case, these tests are not considered to be critical to the accetance of the study as a chronic toxicity feeding study in rats. The NEOL for chronic effects is 250 ppm based upon an increase in hypertrophy/steatosis of the liver (both sexes). The LOEL is 1250 ppm. As an oncogenicity study in rats, the Core Classification is SUPPLEMENTARY because the observed chronic effects are not considered to be severe enough to indicate that the animals were tested at a sufficiently high dose level for an adequate oncogenicity study. The effects seen were not considered to be potentially life threatening and included increased liver weights, cholesterol, hepatic aminopyrene-N-demethylase, and alanine transaminase. No supporting microscopic lesions were observed. One of the criteria for considering whether or not the animals were tested at anadequately high dose level is a significant decrease in body weight and body weight gain. In this study, the females had a significant decrease in body weight and body weight gain. However, as discussed in the food consumption results section, the decrease appears to be related to food consumption because the food concersion efficiency was not affected. Therefore, in this case, the decrease in body weight and body weight gain was not biologically significant.
Delayed neurotoxicity (WHO)
Wistar rats (24 females/dose) were dosed with paclobutrazol (92.4% purity) in corn oil at 0, 2.5, 10, 40 or 100 mg/kg daily from days 7-16 of gestation, inclusive (Killick et al., 1984). No adverse effects were noted in the maternal rats. Abnormalities of the urogenital system were noted in fetuses from the two high dose groups (40 or 100 mg/kg-day). The abnormalities consisted of pelvic dilation of the kidney, and dilation and/or kinking of the ureter. There was a toxicologically significant correlation between effects in the kidney and ureter. Eight fetuses from the high dose group exhibited hydroureter. These findings contrast with the first study (Killick et al., 1983a), in which defects of the kidney and ureter were of low incidence, and not treatment related. The author suggested:”This indicates a change in the background incidence and/or an increased sensitivity of detection” (Pigott, 1984). There was also a significant (P < 0.05) increase in the number of minor skeletal abnormalities indicative of impaired ossification, and extra thoracic ribs at the two highest doses. However, no instances of cleft palate were observed. The NOEL for developmental toxicity (urogenital abnormalities, skeletal abnormalities) was 10 mg/kg-day. This study was acceptable according to FIFRA guidelines.
Hormonal disruption (WHO)
No effect on hormone.
Teratogenicity & Reproduction (WHO)
Alpk/AP rats (15 males/dose, 30 females.dose) were fed continuously for two generations on a diet containing paclobutrazol (92.4% purity) at concentrations of 0, 50, 250, or 1250 ppm (Wickramaratne, 1987). No adverse reproductive effects were noted, so the reproductive NOEL > 1250 ppm (approximately 121 mg/kg-day; base on reported food consumption). Motting, or accentuation of the lobular pattern of the live was observed in 11/30 F0 females, and in F1A, F1B and F2A pups of both sexes in the 1250 ppm group. The parental NOEL for marginal reduction in body weight gain (4%) and hepatic centrilobular steatosis was 250 ppm (approximately 24 mg/kg-day; based on reported food consumption). This study was accetapble according to FIFRA guidelines.
Mutagenicity (WHO)
Paclobutrazol (92.4% purity) was tested on Salmonella strains TA1535, TA1537, TA1538, TA98 and TA100, with and without metabolic (S-9) activation (Callander et al., 1983). Test doses included vihicle (DMSO) and a dose range of 1.6 to 5000 μg/plate, 3 plates/dose and confirmatory assays for all strains. There was no increase in revertant colonies. This study was acceptable to DPR (DPR, 1992). A mammalian cell gene mutation assay was conducted (MacGregor et al., 1983). L5172Y TK+/- Mouse lymphoma cell were exposed for 3 hours in vitro to paclobutrazol (92.4% purity) over a dose range of 1 to 100 μg/ml (first assay) and 60 to 140 μg/ml (second assay), 3 plates/dose, with and without S-9 activation. No increase in the number of colonies was noted. This study was acceptable to DPR.
ECOTOXICOLOGY
- Aquatic Organisms
Fish LC50 (96 h) for rainbow trout 27.8 mg/l. NOEC 3.3 mg/l.
Daphnia LC50 (48 h) 33.2 mg/l.
Algae EC50 (cell volume) for Chlorella fusca 180 mmol/l (Pestic. Sci.,47, 337 (1996)).
2- Earthworms
For Eisenia foetida, acute 14 days, LC50 >5000 mg a.s./kg d.w.soil (mg a.s/ha)
3-Honey bees
Acute oral LD50 >2.0 μg/bee, acute contact LD50 >50 μg/bee.
4- Birds
Acute oral LD50 for mallard ducks >7900 mg/kg, for Japanese quail >2100 mg a.s./kg.
5- Parasites and Predators
The risk was assessed as low for the other non-target organisms for the representative use evaluated.
ENVIRONMENTAL CHEMISTRY
Fate in Soil:
In soil laboratory incubations under aerobic conditions in the dark, paclobutrazol exhibits moderate to very high persistence, forming two major (>10 % app lied radioactivity (AR)) soil metabolites, referred to as CGA149907 (keto form of the parent molecule) and NOA457654. The rate of mineralisation to carbon dioxide varied between 0.2 - 11.1 % AR, while the formation of unextractable residues was a sink, accounting for 4.4 - 52.2 % AR after 84 - 120 days. Paclobutrazol also exhibited slow degradation under anaerobic conditions, forming no novel metabolites. In the study on photolysis in soil, the degradation of paclobutrazol slightly accelerated with production of only minor (≤ 4.2% AR) photo-degradates. Metabolite CGA149907 exhibited moderate to high persistence, and metabolite NOA457654 exhibited low to modera te persistence in soil. Dissipation of paclobutrazol was investigated in a number of field trials, which confirmed the relatively slow disappearance of paclobutrazol from the soil compartment. Paclobutrazol exhibits very high to low mobility in soil. Metabolite CGA149907 exhibits medium to low mobility, while the metabolite NOA457654 exhibits very high mobility in soil. There was an indication that the adsorption of CGA149907 is pH dependent. The soil plateau concentration fo r long-term use in consecutive years and PECsoil (predicted environmental concentration) for pacl obutrazol was calculated based on the worst-case non-normalized field DT50. For the metabolites (CGA149907, NOA457654 and also for 1,2,4-triazole), initial PECsoil values were calculated based on the initial PECsoil of paclobutrazol.
Effect on microbial Process:
Nitrogen mineralisation
Up to 77 days, 0% effect at any sample days at 8.0 mg a.s./kg d.w.soil (5.0 kg a.s./ha)
Carbon mineralisation
0% effects by the end of the study at 20 kg a.s./ha in 21 days.
Method of Res.Analysis: GC/HPLC
Fate in Plant:
Paclobutrazol is readily taken up through roots as well as stems and foliage and is translocated almost exclusively in the xylem.
In two studies on apples harvested from trees sprayed with paclobutrazol, labelled in either the phenyl or triazolyl group, paclobutrazol itself was the major component (about 50%) of the residue, most of which was in the peel. Other compounds identified were the diol, the acid and triazollalanie, although there were significant differences between the results of the two sudies. In view of the comments in the 1987 JMPR report on the toxicological significance of residues of triazolylalanine the meeting agreed to defer further comments until a definitive metabolism study in apples has been carried out and toxicological studies on triazolylalanine have been evaluated.
Fate in Water:
Paclobutrazol is stable to hydrolysis and it was stab le in a standard aqueous photolysis study. In a photolysis study using natural pond water, paclobutrazol exhibited slow degradation, forming two major peaks that contained multiple components. Paclobutrazol is not readily biodegradable. In laboratory incubations in aerobic natural sediment water systems, paclobutrazol exhibited very high persistence (estimated half-life 193 to >1000 days), forming no major metabolites.
Photo degradation Rate:
In the study on photolysis in soil, the degradation of paclobutrazol slightly accelerated with production of only minor (≤ 4.2% AR) photo-degradates.
In a photolysis study using natural pond water, paclobut razol exhibited slow degradation, forming two major peaks that contained multiple components.
Rate of hydrolysis at PH levels :
No degradation during a 30 day study at 25oC at pH 4, 7 and 9. Paclobutrazol considered stable to hydrolysis.
Methods of analysis: GC/HPLC
Fate in Animal:
Approximately 95% of an oral dose of paclobutrazol was absorbed by rats. An average of 60% of the rats' oral dose was excreted in the urine, and 35% in the feces. Virtally all paclobutrazol, regardless of dose, was cleared within 168 hours. Paclobutrazol was converted in rats to diol and carboxytic acid metabolites, which were excreted both conjugated and unconjugated in the urine and bile. The highest tissue concentrations were found in the liver and gastrointestinal system, but paclobutrazol did not bioconcentrate in tissue. Radiolabelled paclobutrazol was absorbed dermally by rats at rates of between 5 to 28% at 24 hours.
SAFETY HANDLING STORAGE AND DISPOSAL
Storage:
Requirements for storage areas and containers
Keep out of the reach of children.
Advice on common storage
Keep away from food, drink and animal feedingstuffs.
Suitable materials
Rigid material with protection against moisture.
Shelf Life:
2 years.
Handling Precautions:
Do not breathe dust. Avoid contact with eyes, skin, or clothing. Wash thoroughly after handling. Wash clothing after use. Do not store or consume food, drink, or tobacco in areas where they may become contaminated with this material.
Containers (Lining):
100 mL, 250 mL, 500 mL, 1 L plastic bottle, 200 mL drum. Or other packing according to requirement.
Signs and symptoms of Over Exposure:
Dizziness, headache, nausea, abdomen convulsion, vomit, hidrosis, myosis, weep, spit hypersecretion, can cause lethal danger if contact or inhale to much directly.
Note to Physician:
No special advice. Treat symptomatically.
FIRST AID:
IF Swallowed:
If the victim is convulsing or unconscious, administer a glass of water, ensure that the victim's airway is open and if symptoms developed, call a physician immediately.
If in eyes:
Hold eye open and rinse slowly and gently with water for 15 to 20 minutes. Remove contact lenses, if present, after the first 5 minutes, then continue rinsing eye. Call a poison control center or doctor for treatment advice.
If on skin or clothing: Take off contaminated clothing. Rinse skin immediately with plenty of water for 15-20 minutes. Call a poison control center or doctor for treatment advice.
IF Inhaled
If person is not breathing, call an ambulance, then give artificial respiration, preferably mouth-to-mouth if possible. Call a poison control center or doctor for further treatment advice. Take affected person to fresh air.
ANTIDOTE:
No special antidote.
Administer glasses of water if swallow.
REMARKS:
The information above is believed to be accurate and represents the best information currently available to us. However, we make no warranty of merchant ability or any other warranty, express or implied, with respect to such information, we assumes no legal responsibility for use or reliance upon these data. The date on this sheet relates only to the specific material designated herein, and users should make their own investigations to determine the suitability of the information for their particular purposes.
|





